 Each hydrogen requires one covalent bond to fill its first shell. In a methane molecule, carbon effectively has a “full” second shell (eight electrons) and each hydrogen has a “full” first shell (two electrons). The carbon atom forms a covalent bond with four hydrogen atoms to form a molecule of methane. Hydrogen has a single electron in its outermost shell and can share this electron with one other atom. The outer shell of carbon has four electrons, so carbon can share an electron with four other atoms (which will then give carbon a full outer shell of 8 electrons). One carbon atom and four hydrogen atoms react to form methane. One example of covalent bonding to form a molecule is the formation of methane, a colorless and flammable gas that results from burning gasoline or fossil fuels (Fig. Covalent bonds are the strongest, most stable types of chemical bonds in the biological world. This sharing of electrons is called a covalent bond. Atoms can also share two pairs of electrons (in a double bond). A single pair of shared electrons makes a single covalent bond. In essence, a shared electron is counted “twice” and participates in a larger shell that joins the two atoms. Instead of transferring their electrons completely, atoms typically remain in very close contact and share electrons so that their outer shells are filled. hydrogen, carbon, nitrogen, and oxygen) are considered stable with eight electrons in this outer shell. While some elements may be able to hold more electrons in their third shell, most of the important elements in biology (e.g. Atoms tend to bond to other atoms in such a way that both atoms have filled outer shells as a result of the interaction. The first electron shell of an atom is considered full (or stable) when it contains two electrons, and the second and third shells are full (stable) with eight electrons. It is not easy to explain why this is true, but it’s a rule of thumb that predicts how atoms will react with each other. The most stable situation for an atom is to have its outer shell completely filled with electrons. By bonding, electrons find pairs, and atoms become part of a molecule. A chemical bond is the attractive force between atoms and contains energy. In most cases, these unpaired electrons are used to create chemical bonds. An atom with an odd number of electrons must have an unpaired electron. Atoms are more stable when their electrons orbit in pairs. However, this energy is not always stable, depending on the number of electrons within an atom. This energy is stored within the charge and movement of electrons and the bonds that atoms make with one another. Molecules are composed of two or more atoms held together by chemical bonds. Molecules and Covalent BondingĪtoms combine to form a larger and more complex entity called a molecule. 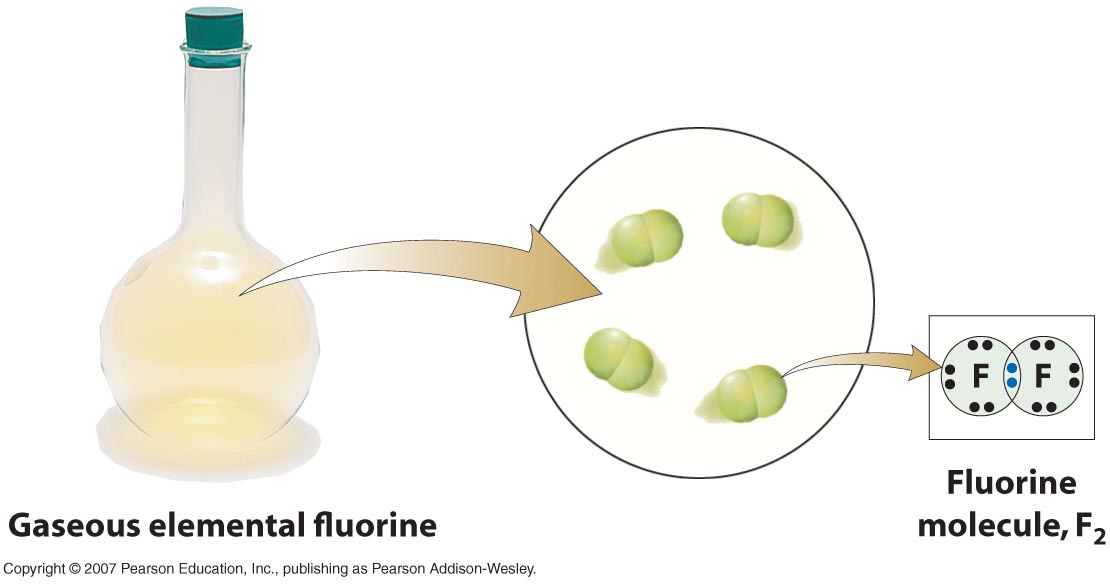
Protons and neutrons are found in the nucleus (center) of the atom. Diagram of a lithium atom, showing the placement of protons, neutrons, and electrons. 
This attraction keeps the atom together, much like the force of gravity keeps the moon in orbit around Earth.įigure 1.3. And since opposites attract, electrons are attracted to this nucleus and move around it in an electron cloud surrounding the nucleus. Because protons and neutrons are contained in the dense nucleus of the atom, the nucleus has a positive charge. 
Protons have a positive charge, neutrons have no charge, and electrons are negatively charged. Protons and neutrons are found in the nucleus (center) of the atom, while electrons are found outside the nucleus in regions called shells. The atoms themselves are made of even smaller particles called protons, neutrons, and electrons. Even within a single microscopic cell, there is room for not just billions, but trillions or even hundreds of trillions of atoms. These four elements make up the bulk of all living things.Ītoms are unimaginably small. Note the four elements circled in blue (hydrogen, carbon, nitrogen, and oxygen). 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
